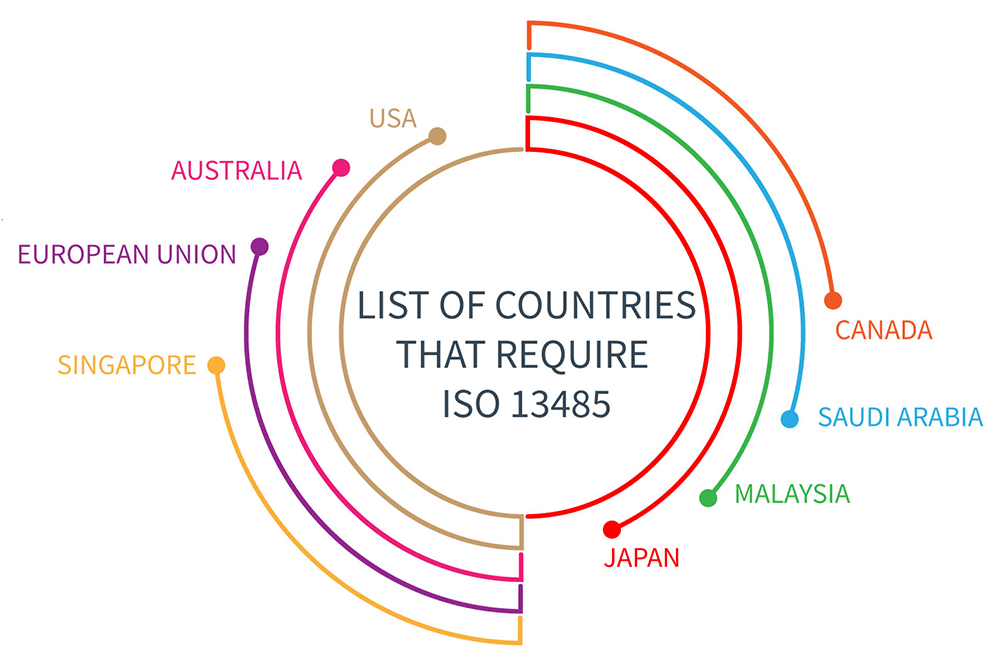
#Iso 13485 certification iso
If you wish to get ISO 13485 certification, please send us an enquiry. Enhancing trust of public and public monitoring bodies in respect of the medical devices manufacturer.Optimization of costs reduction of operating costs, reduction of nonconforming products costs, raw materials, energy and other resources savings, ISO 13485 potentially beneficial for medical device organizations as it: Enhances the organizations ability to meet customers and regulatory requirements.Attestation of efficiency and effectiveness of the established quality management system by an independent third party.Simplifying the process of attestation of conformity to requirements of EU directives and corresponding Government Orders.Guarantee of production process stability and thus steady and high quality of services and products supplied to customers.Decision on withdrawal of the certificate in case of fundamental deviations from requirements of standards.Decision on suspension of the certificate ISO 13485 contains requirements that are essential for any organisation operating at any tier in the medical device and pharmaceutical supply chain, including.Decision on confirmation of the certificate validity until the next surveillance.One of the following is issued depending on the surveillance findings Assessment of the Audit Report by the Certification Bodyĭuring the three year validity period of the certificate, a surveillance audit takes place once a year.Elaboration of the Certification Audit Report.Stage II: On-site evaluation of quality management system.
#Iso 13485 certification verification

The certification process consists of 3 basic phases:ģ) Certification by an accredited certification body The standard contains the requirement for quality management systems, to meet customer requirement and incorporate applicable regulatory requirements within an organization’s quality management system. ISO 13485 is applicable to designers, manufacturers, and distributors of medical devices. ISO 13485 is Quality Management System for Medical Devices.
